 Winston Liu, a Montgomery Blair High School student and intern Carnegie, has been named a semifinalist in the Siemens Foundation’s 2010 competition in Math, Science, and Technology. Liu's study on “Ethane-hydrogen systems under high pressure” is an energy-related project which helps clarify phase behavior in the hydrogen-hydrocarbon family of systems to 7.5 GPa. He has been working on under the mentorship of Jinfu Shu and Ho-kwang Mao.
Winston Liu, a Montgomery Blair High School student and intern Carnegie, has been named a semifinalist in the Siemens Foundation’s 2010 competition in Math, Science, and Technology. Liu's study on “Ethane-hydrogen systems under high pressure” is an energy-related project which helps clarify phase behavior in the hydrogen-hydrocarbon family of systems to 7.5 GPa. He has been working on under the mentorship of Jinfu Shu and Ho-kwang Mao. Liu puts his research in light of growing environmental and energy problems in the world which can only be solved through a gradual shift from hydrocarbon sources to clean, renewable sources such as hydrogen. Research on compressed hydrogen-hydrocarbon systems is of fundamental importance for a variety of applications spanning from reactions within planetary interiors to energy storage.
A Raman study of the hydrocarbon ethane and hydrogen was conducted in diamond anvil cells to 7.5 GPa at 300K. Compositions of 1:1, 3:1, and 12:1 C2H6:H2 %mol were studied. The interaction between the two has been characterized as a simple binary eutectic system and a preliminary phase diagram is presented. Raman spectra for the system to 7.5 GPa revealed pure H2 and C2H6 crystals, and potential metastability. In addition, two new Raman peaks for ethane at ~2770 and ~2740 wavenumbers at ambient pressure were observed. Research on this important system will lay a foundation for further research in the new field of high-pressure chemistry.
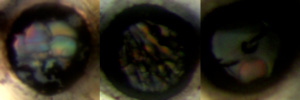
Image Caption: Photomicrographs of C2H6 + H2 samples. All holes pictured have diameters of 4006 microns. (left) 73% H2 sample at 5.1 GPa; crystals pictured are ethane crystals. (center) 93% H2 sample at 6.7 GPa; crystals pictured are hydrogen crystals. (right) 73% H2 sample at 7.2 GPa after cell was heated and then cooled to allow crystal growth and development. After hitting the eutectic point at 7.1 GPa, cell was originally nearly opaque with micro-crystals.
